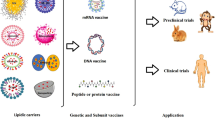
Abstract
Lipid-based carriers, including liposomes and emulsions, have been studied as antigen delivery systems to improve humoral and cellular immune responses. Lipid-based delivery systems have been tailored based on the physicochemical properties of antigens and administration routes. Non-covalent encapsulation or chemical conjugation of antigens onto the surface of nanocarriers has been reported to load high amounts of antigen, protect antigens from degradation, and enhance cellular uptake by immune cells. Moreover, active targeting to antigen-presenting cells can be achieved through surface modification of lipid-based nanostructures. Antigens, which include proteins, peptides and nucleic acids, have been delivered by appropriate lipid-based delivery systems, and antigen-loaded lipid nanoparticles have been shown to cause greater immune responses compared with naked antigens. For in vivo applications, both invasive and non-invasive routes have been adopted for vaccination with antigen-loaded lipid nanocarriers. In this review, we address recent progress, current status, and clinical applications of lipid-based antigen delivery systems.


Similar content being viewed by others
References
Alving CR, Beck Z, Matyas GR, Rao M (2016) Liposomal adjuvants for human vaccines. Expert Opin Drug Deliv 25:1–10
Barnier-Quer C, Elsharkawy A, Romeijn S, Kros A, Jiskoot W (2013) Adjuvant effect of cationic liposomes for subunit influenza vaccine: influence of antigen loading method, cholesterol and immune modulators. Pharmaceutics 5(3):392–410
Barouch DH (2008) Challenges in the development of an HIV-1 vaccine. Nature 455:613–619
Bioley G, Lassus A, Bussat P, Terrettaz J, Tranquart F, Corthésy B (2012) Gas-filled microbubble-mediated delivery of antigen and the induction of immune responses. Biomaterials 33:5935–5946
Bioley G, Zehn D, Lassus A, Terrettaz J, Tranquart F, Corthésy B (2013) The effect of vaccines based on ovalbumin coupled to gas-filled microbubbles for reducing infection by ovalbumin-expressing Listeria monocytogenes. Biomaterials 34:5423–5430
Bogers WM, Oostermeijer H, Mooij P, Koopman G, Verschoor EJ, Davis D, Ulmer JB, Brito LA, Cu Y, Banerjee K, Otten GR, Burke B, Dey A, Heeney JL, Shen X, Tomaras GD, Labranche C, Montefiori DC, Liao HX, Haynes B, Geall AJ, Barnett SW (2015) Potent immune responses in rhesus macaques induced by nonviral delivery of a self-amplifying RNA vaccine expressing HIV type 1 envelope with a cationic nanoemulsion. J Infect Dis 211:947–955
Bovier PA (2008) Epaxal: a virosomal vaccine to prevent hepatitis A infection. Expert Rev Vaccines 7:1141–1150
Brito LA, Chan M, Shaw CA, Hekele A, Carsillo T, Schaefer M, Archer J, Seubert A, Otten GR, Beard CW, Dey AK, Lilja A, Valiante NM, Mason PW, Mandl CW, Barnett SW, Dormitzer PR, Ulmer JB, Singh M, O’Hagan DT, Geall AJ (2014) A cationic nanoemulsion for the delivery of next-generation RNA vaccines. Mol Ther 22:2118–2129
Carneiro C, Correia A, Collins T, Vilanova M, Pais C, Gomes AC, Real Oliveira ME, Sampaio P (2015) DODAB:monoolein liposomes containing Candida albicans cell wall surface proteins: a novel adjuvant and delivery system. Eur J Pharm Biopharm 89:190–200
Chavoshian O, Biari N, Badiee A, Khamesipour A, Abbasi A, Saberi Z, Jalali SA, Jaafari MR (2013) Sphingomyelin liposomes containing soluble Leishmania major antigens induced strong Th2 immune response in BALB/c mice. Iran J Basic Med Sci 16(9):965–972
Chen L, Zhu J, Li Y, Lu J, Gao L, Xu H, Fan M, Yang X (2013) Enhanced nasal mucosal delivery and immunogenicity of anti-caries DNA vaccine through incorporation of anionic liposomes in chitosan/DNA complexes. PLoS One 8(8):e71953
D’Acremont V, Herzog C, Genton B (2006) Immunogenicity and safety of a virosomal hepatitis A vaccine (Epaxal) in the elderly. J Travel Med 13:78–83
Eskandari F, Talesh GA, Parooie M, Jaafari MR, Khamesipour A, Saberi Z, Abbasi A, Badiee A (2014) immunoliposomes containing soluble Leishmania antigens (SLA) as a novel antigen delivery system in murine model of leishmaniasis. Exp Parasitol 146:78–86
Fan Y, Sahdev P, Ochyl LJ, Akerberg J, Moon JJ (2015) Cationic liposome-hyaluronic acid hybrid nanoparticles for intranasal vaccination with subunit antigens. J Control Release 208:121–129
Firouzmand H, Badiee A, Khamesipour A, Heravi Shargh V, Alavizadeh SH, Abbasi A, Jaafari MR (2013) Induction of protection against leishmaniasis in susceptible BALB/c mice using simple DOTAP cationic nanoliposomes containing soluble Leishmania antigen (SLA). Acta Trop 128:528–535
Ghaffar KA, Giddam AK, Zaman M, Skwarczynski M, Toth I (2014) Liposomes as nanovaccine delivery systems. Curr Top Med Chem 14:1194–1208
Giddam AK, Zaman M, Skwarczynski M, Toth I (2012) Liposome-based delivery system for vaccine candidates: constructing an effective formulation. Nanomedicine (Lond) 7:1877–1893
Glück R, Mischler R, Brantschen S, Just M, Althaus B, Cryz SJ Jr (1992) Immunopotentiating reconstituted influenza virus virosome vaccine delivery system for immunization against hepatitis A. J Clin Invest 90:2491–2495
Herzog C, Hartmann K, Künzi V, Kürsteiner O, Mischler R, Lazar H, Glück R (2009) Eleven years of Inflexal V-a virosomal adjuvanted influenza vaccine. Vaccine 27:4381–4387
Hjálmsdóttir Á, Bühler C, Vonwil V, Roveri M, Håkerud M, Wäckerle-Men Y, Gander B, Johansen P (2016) Cytosolic delivery of liposomal vaccines by means of the concomitant photosensitization of phagosomes. Mol Pharm. doi:10.1021/acs.molpharmaceut.5b00394
Hu Y, Zheng H, Huang W, Zhang CA (2014) Novel and efficient nicotine vaccine using nano-lipoplex as a delivery vehicle. Hum Vaccin Immunother 10(1):64–72
Kallert S, Zenk SF, Walther P, Grieshober M, Weil T, Stenger S (2015) Liposomal delivery of lipoarabinomannan triggers Mycobacterium tuberculosis specific T-cells. Tuberculosis (Edinb). 95:452–462
Lanzi A, Fehres CM, de Gruijl TD, van Kooyk Y, Mastrobattista E (2014) Effects of antigen-expressing immunostimulatory liposomes on chemotaxis and maturation of dendritic cells in vitro and in human skin explants. Pharm Res 31(2):516–526
Li P, Chen S, Jiang Y, Jiang J, Zhang Z, Sun X (2013) Dendritic cell targeted liposomes-protamine-DNA complexes mediated by synthetic mannosylated cholesterol as a potential carrier for DNA vaccine. Nanotechnology 24:295101
Malik B, Gupta RK, Rath G, Goyal AK (2014) Development of pH responsive novel emulsion adjuvant for oral immunization and in vivo evaluation. Eur J Pharm Biopharm 87:589–597
Mischler R, Metcalfe IC (2002) Inflexal V a trivalent virosome subunit influenza vaccine: production. Vaccine 20:B17–B23
Sahdev P, Ochyl LJ, Moon JJ (2014) Biomaterials for nanoparticle vaccine delivery systems. Pharm Res 31:2563–2582
Schwendener RA (2014) Liposomes as vaccine delivery systems: a review of the recent advances. Ther Adv Vaccines 2:159–182
Senchi K, Matsunaga S, Hasegawa H, Kimura H, Ryo A (2013) Development of oligomannose-coated liposome-based nasal vaccine against human parainfluenza virus type 3. Front Microbiol 4:346
Smith DM, Simon JK, Baker JR Jr (2013) Applications of nanotechnology for immunology. Nat Rev Immunol 13:592–605
Takagi A, Kobayashi N, Taneichi M, Uchida T, Akatsuka T (2013) Coupling to the surface of liposomes alters the immunogenicity of hepatitis C virus-derived peptides and confers sterile immunity. Biochem Biophys Res Commun 430:183–189
Trentini MM, de Oliveira FM, Gaeti MP, Batista AC, Lima EM, Kipnis A, Junqueira-Kipnis AP (2014) Microstructured liposome subunit vaccines reduce lung inflammation and bacterial load after Mycobacterium tuberculosis infection. Vaccine 32(34):4324–4332
Tyagi RK, Garg NK, Jadon R, Sahu T, Katare OP, Dalai SK, Awasthi A, Marepally SK (2015) Elastic liposome-mediated transdermal immunization enhanced the immunogenicity of P. falciparum surface antigen, MSP-119. Vaccine 33:4630–4638
Usonis V, Bakasénas V, Valentelis R, Katiliene G, Vidzeniene D, Herzog C (2003) Antibody titres after primary and booster vaccination of infants and young children with a virosomal hepatitis A vaccine (Epaxal). Vaccine 21:4588–4592
Wang HW, Jiang PL, Lin SF, Lin HJ, Ou KL, Deng WP, Lee LW, Huang YY, Liang PH, Liu DZ (2013) Application of galactose-modified liposomes as a potent antigen presenting cell targeted carrier for intranasal immunization. Acta Biomater 9:5681–5688
Wang N, Wang T, Zhang M, Chen R, Niu R, Deng Y (2014a) Mannose derivative and lipid A dually decorated cationic liposomes as an effective cold chain free oral mucosal vaccine adjuvant-delivery system. Eur J Pharm Biopharm 88:194–206
Wang N, Wang T, Zhang M, Chen R, Deng Y (2014b) Using procedure of emulsification-lyophilization to form lipid A-incorporating cochleates as an effective oral mucosal vaccine adjuvant-delivery system (VADS). Int J Pharm 468:39–49
Watarai S, Iwase T, Tajima T, Yuba E, Kono K (2013) Efficiency of pH-sensitive fusogenic polymer-modified liposomes as a vaccine carrier. Sci World J 2013:903234
Wilschut J (2009) Influenza vaccines: the virosome concept. Immunol Lett 21(122):118–121
Woelbing F, Kostka SL, Moelle K, Belkaid Y, Sunderkoetter C, Verbeek S, Waisman A, Nigg AP, Knop J, Udey MC, Stebut E (2006) Uptake of Leishmania major by dendritic cells is mediated by Fcγ receptors and facilitates acquisition of protective immunity. J Exp Med 203:177–188
Wong PT, Wang SH, Ciotti S, Makidon PE, Smith DM, Fan Y, Schuler CF 4th, Baker JR Jr (2014) Formulation and characterization of nanoemulsion intranasal adjuvants: effects of surfactant composition on mucoadhesion and immunogenicity. Mol Pharm 11:531–544
Zhen Y, Wang N, Gao Z, Ma X, Wei B, Deng Y, Wang T (2015) Multifunctional liposomes constituting microneedles induced robust systemic and mucosal immunoresponses against the loaded antigens via oral mucosal vaccination. Vaccine 33:4330–4340
Acknowledgments
This work has been financially supported by a Grant from the Korean Health Technology R&D Project (No. HI15C2842), Ministry of Health & Welfare, Republic of Korea. These authors (JY Park, G Shim, MG Kim, and YK Oh) declare that they have no conflict of interest. This article does not contain any studies with human and animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Park, J.Y., Kim, MG., Shim, G. et al. Lipid-based antigen delivery systems. Journal of Pharmaceutical Investigation 46, 295–304 (2016). https://doi.org/10.1007/s40005-016-0246-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-016-0246-z